Fluvoxamine Covid Clinical Trial
In the meantime evidence was coming in from. 21 Recent evidence indicates a potential therapeutic role of fluvoxamine for COVID-19.
On April 23 2021 fluvoxamine was added in the US National Institutes of Health NIH COVID-19 Guidelines Panel although there is insufficient evidence for the efficacy of fluvoxamine.

Fluvoxamine covid clinical trial. Fluvoxamine vs placebo and clinical deterioration in outpatients with symptomatic COVID-19. 2292-2300 View in Article. The STOP-COVID 2 study was a decentralized hub-and-spoke clinical trial.
The fluvoxamine arm of the trial began in January 2021 and involved around 1400 Brazilian patients who were seen for covid-19 as outpatients at an emergency room all evaluated to be high risk. This did not result in a reduction in hospitalizations or mortality although the study was not powered for the latter. The TOGETHER randomised platform clinical trial.
Washington University is currently evaluating whether to open a clinical trial for treating people with fluvoxamine for long-haul COVID. This study is fully-remote which means that there is no face-to-face contact. 2021 Effect of early treatment with fluvoxamine on risk of emergency care and hospitalisation among patients with COVID-19.
Mattar began the preliminary pragmatic non-contact but high-touch STOP COVID trial in April 2020. Placebo in preventing either extended emergency room observation or hospitalization due to COVID. PREPRINT August 23 2021.
This clinical trial aims to determine if fluvoxamine a selective serotonin reuptake inhibitor with high affinity for the sigma-1 receptor can be used in mild to moderate COVID-19 to prevent the progression to severe COVID-19. Fluvoxamine remains an interesting repurposed medication for COVID-19 however the only outcome of significance was reduced ED visits 6hrs. Fluvoxamine is an anti-depressant drug approved by the FDA for the treatment of obsessive-compulsive disorder and has a potential for immune modulation as a sigma-1.
There is insufficient evidence for the COVID-19 Treatment Guidelines Panel to recommend either for or against the use of fluvoxamine for the treatment of COVID-19. Reiersen and Lenze started recruiting for a larger-scale trial looking for more evidence that fluvoxamine could be a first-line Covid-19 treatment. Results from the preliminary study showed that 0 of patients experienced clinical deterioration in the Fluvoxamine group while 83 of patients in the placebo group deteriorated.
27 UPI --The anti-depressant drug fluvoxamine reduces the risk for prolonged hospital stays in people with severe COVID-19 a clinical trial published Wednesday by. A randomized clinical trial. Ivermectin Fluvoxamine Physicians who had been prescribing ivermectin plus supplements have reported markedly superior outcomes when adding fluvoxamine.
Placebo in preventing either extended emergency room observation or hospitalization due to COVID-19. The use of fluvoxamine for the treatment of COVID-19 is considered investigational which means the US Food and Drug Administration has not approved it for this use. Gilmar Reis Eduardo Augusto dos Santos Moreira-Silva Daniela Carla.
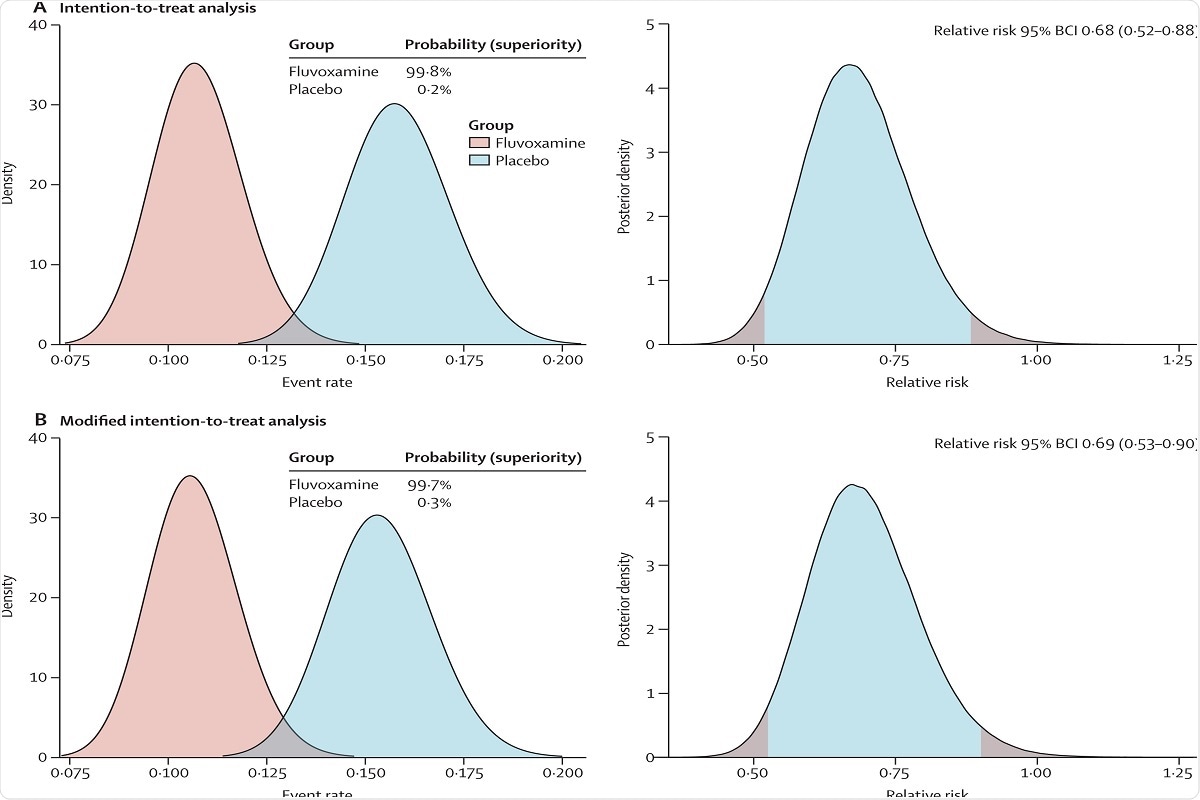
In the TOGETHER randomized platform clinical trial for acutely symptomatic patients with COVID-19 we assessed the efficacy of fluvoxamine vs. This arm of the TOGETHER trial is the largest randomized trial. On August 6 2021 the interim results of TOGETHER trial typeclinical-trialattrstextNCT04727424term_idNCT04727424 NCT04727424 by a multinational.
In this randomized trial that included 152 adult outpatients with confirmed COVID-19 and symptom onset within 7 days clinical deterioration occurred in 0 patients treated with fluvoxamine vs 6 83 patients treated with placebo over. The results of a new randomized clinical trial published in The Lancet Global Health have demonstrated that using fluvoxamine to treat high-risk outpatients with early-diagnosed COVID-19 reduced the need for prolonged observation in an emergency setting or hospitalization compared to a control group who received a placebo. Background Recent evidence indicates a potential therapeutic role of fluvoxamine for COVID-19In the TOGETHER randomized platform clinical trial for acutely symptomatic patients with COVID-19 we assessed the efficacy of fluvoxamine vs.
Background Recent evidence indicates a potential therapeutic role of fluvoxamine for COVID-19. Placebo in preventing either extended emergency 24 room observation or hospitalization due to COVID-19. Results from adequately powered well-designed and well-conducted clinical trials are needed to provide more specific evidence-based guidance on the role of fluvoxamine for the treatment of COVID-19.
In the 22 TOGETHER randomized platform clinical trial for acutely symptomatic patients with COVID-19 23 we assessed the efficacy of fluvoxamine vs. Study materials including study drug will be shipped to participants houses. The TOGETHER randomised platform clinical trial.
And the patients had a higher rate of hospitalization than Covid-19 patients in other clinical trials. By Gilmar Reis et al. Effect of early treatment with fluvoxamine on risk of emergency care and hospitalisation among patients with.
Reierson and colleagues gave 741 volunteers with Covid-19 100 mg of fluvoxamine twice a.

Fluvoxamine Helps In Reducing Extended Hospitalization Study
Covid Fluvoxamine The Antidepressant Likely To Cut Down Hospitalization Risks A Study Found Sortiraparis Com

Covid 19 Trial Conducted By Mail Tests Drug To Prevent Hospitalization

Fluvoxamine May Prevent Severe Covid 19 Onset Suggests Trial
Common Antidepressant Shows Promise In Halting Covid 19 Symptoms Early

.jpg)